Battery Basics
How do Batteries Work?
There’s lots of physics and chemistry going on inside a battery, but we’ll spare you the nerdy details and explain what’s happening in a very basic and simplistic way.
There are three basic components in a battery that we’re going to focus on: the two electrodes, and the separator. The electrodes are the biggest players – they’re responsible for housing all the charge a battery holds, and also for sending that charge to all of your electronics. The separator keeps the two electrodes…separated! We don’t want them touching, that’s a guaranteed fire.
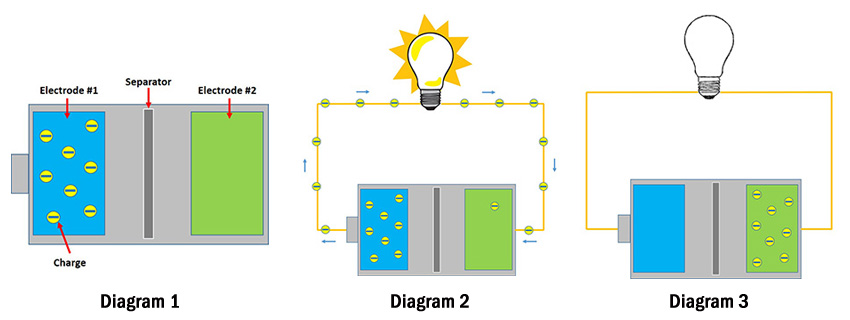
So, in Diagram 1 the blue electrode is housing all the charge; however, the charge really wants to go over to the green electrode. It can’t just cut across because the separator is there, so it has to take the long way. So, what’s a charge to do?
When you connect your battery to something, you’re creating a pathway for the charge to get to the other side (Diagram 2). When the charge travels through your device en-route to the other side, it powers your device. Take the flashlight – when the charge travels from one side of the flashlight’s battery to the other, it has to pass through the light bulb. When it passes through the light bulb, the bulb gives off light (duh…)
Once all the charge in the blue electrode travels over to the green electrode, your battery is considered “empty”(Diagram 3). Some batteries are rechargeable – we call these secondary batteries. With secondary batteries, you plug them into a charger and the whole process reverses itself – all the charge in the green electrode travels back to the blue electrode. When that’s done, your battery is once again full.
Other batteries are non-rechargeable – we call those primary batteries. Think of all those AA batteries you buy for your flashlights and TV remotes. Once they’re empty, there’s no recharging them (unless you want to start a fire). Whether a battery is primary or secondary all depends on the type of battery you’re using, but that’s a discussion for another day.









Leave a reply
You must be logged in to post a comment.